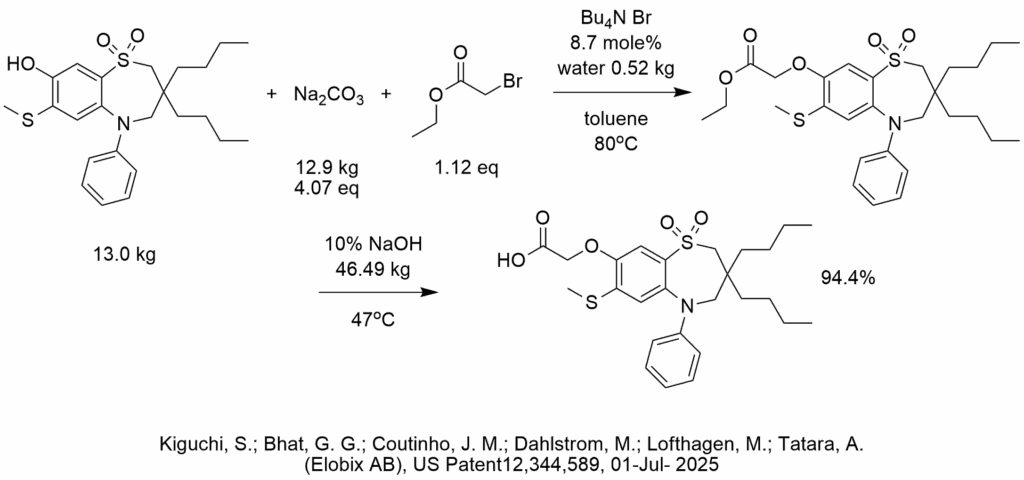
Example 1 in this patent serves as a reminder that phase-transfer catalysis is a workhorse for common straightforward reactions that need to be performed in high yield when working on pilot plant scale (or larger). The procedure also hints at the importance of choosing the right amount of water for a solid-liquid PTC reaction.
In this example, two consecutive PTC reactions are performed in high yield on a 13 kg scale without isolation of the intermediate.
The first reaction is a solid-liquid PTC etherification of a phenol with ethyl bromoacetate, using tetrabutylammonium bromide as the phase-transfer catalyst and sodium carbonate as the base. A small amount of water was added to the etherification reaction. The second step was hydrolysis using a large amount of 10% NaOH that obviously contains a lot of water and performed without isolating the ester intermediate.
One particularly interesting aspect of this solid-liquid PTC etherification was the use of a measured amount of water which in this specific case was 4.0% relative to sodium carbonate base (solid).
As we teach in our 2-day course, Industrial Phase-Transfer Catalysis, there is an optimal amount of water for most solid-liquid PTC systems that should be identified and used to achieve highest reactivity and sometimes highest selectivity. Depending on the specific solid salt being used and other factors, the optimal amount of water can be as low as a few ppm to as high as several tens of percent. In one solid-liquid PTC reaction described in the course, the optimal amount of water in an N-alkylation using potassium carbonate as base was 16 wt% relative to the potassium carbonate. As noted earlier, this particular reaction using sodium carbonate was reported at only one water level which was 4.0%.
It is not known if the inventors of US Patent 12,344,589 studied and optimized the water level for this system, but we speculate that that they were likely aware of the importance of choosing a specific amount of water given that they added 0.52 kg of water after adding 12.89 kg sodium carbonate.
The optimum amount of water for every solid-liquid PTC reaction is different and depends on multiple parameters including the identity of the reactants, especially the specific cation and anion of the salt and its morphology, as well as solvent, phase-transfer catalyst, possible co-catalyst, leaving group and other factors.
The magnitude of the sensitivity of solid-liquid PTC systems toward hydration, often in narrow a range, is often overlooked when developing and commercializing new solid-liquid PTC processes. I have seen commercial PTC processes that produced product for many years and those in charge of plant operations were not aware how far they were from higher profit due to reactivity and/or selectivity, sometimes because the optimum level of hydration is in a narrow range that could easily be missed during development and optimization.
When you are developing a solid-liquid PTC process or optimizing an existing solid-liquid PTC process, you can often improve your process performance AND save weeks or months of process R&D or process optimization time by working with Marc Halpern of PTC Organics to benefit from decades of highly specialized expertise in industrial solid-liquid PTC systems. The ROI on PTC Process Consulting is high when accounting for both more profitable PTC processes and not wasting constrained and expensive process R&D resources. Now contact Marc Halpern of PTC Organics if your company is developing, optimizing or considering developing a commercial process that uses or may use solid-liquid PTC conditions.