 When quat salts appear in the patent literature, they are mostly as phase-transfer catalysts. There are other applications, such as in toners, as electrolytes in specialty batteries with organic solvents and in a variety of other applications. One of the rarer applications of quat salts is as an electrolyte for electrochemical oxidations for organic synthesis.
When quat salts appear in the patent literature, they are mostly as phase-transfer catalysts. There are other applications, such as in toners, as electrolytes in specialty batteries with organic solvents and in a variety of other applications. One of the rarer applications of quat salts is as an electrolyte for electrochemical oxidations for organic synthesis.
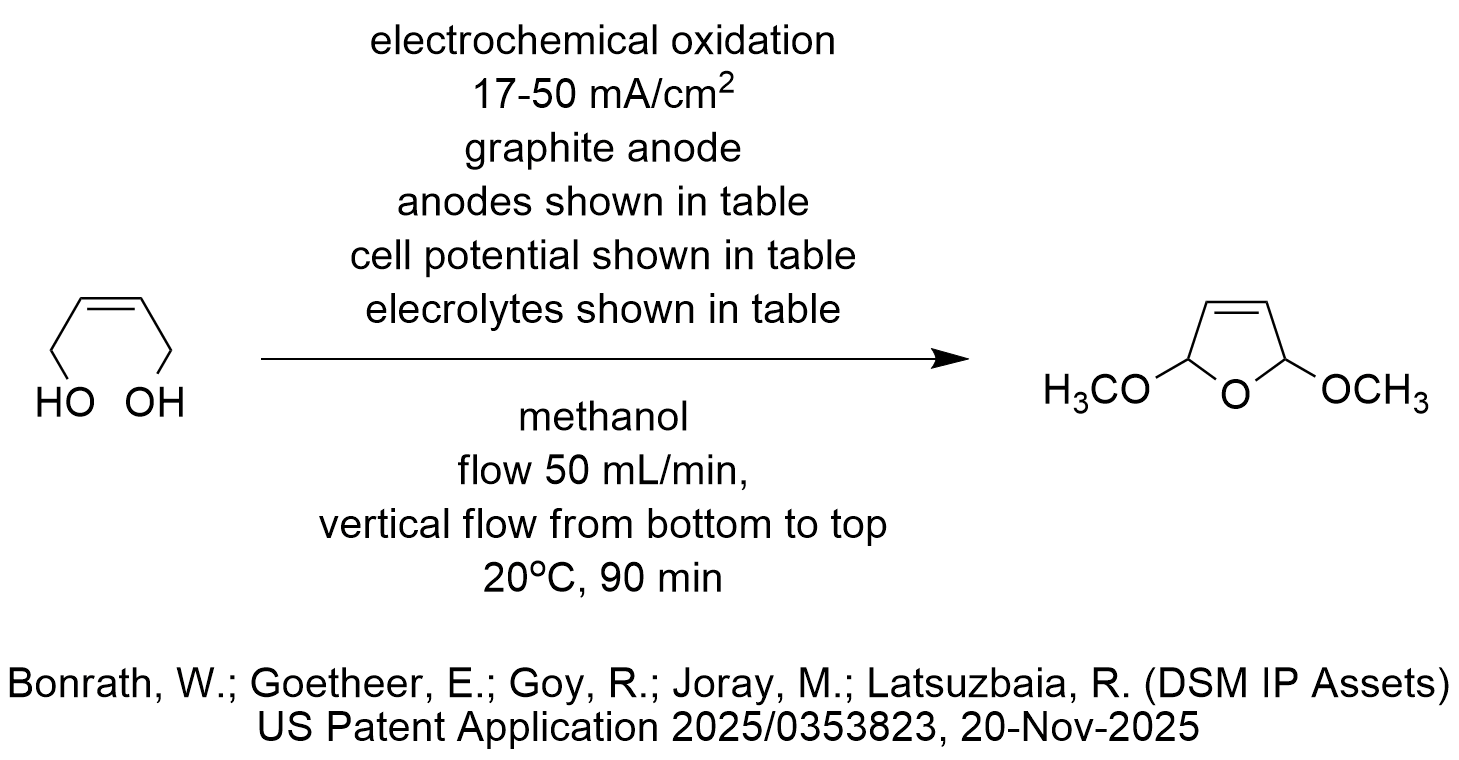
The diagram shows a useful electrochemical oxidation. The substrate is butene-1,4-diol, and the product is 2,5-dimethoxy-2,5-dihydrofuran (DMDF). The oxidation is performed under mild conditions and uses methanol as solvent (and reactant), graphite anode, a flow-through electrochemical cell that feeds the substrate vertically from the bottom flowing to the top at a rate of 50 mL/min and is performed at room temperature for 90 minutes.
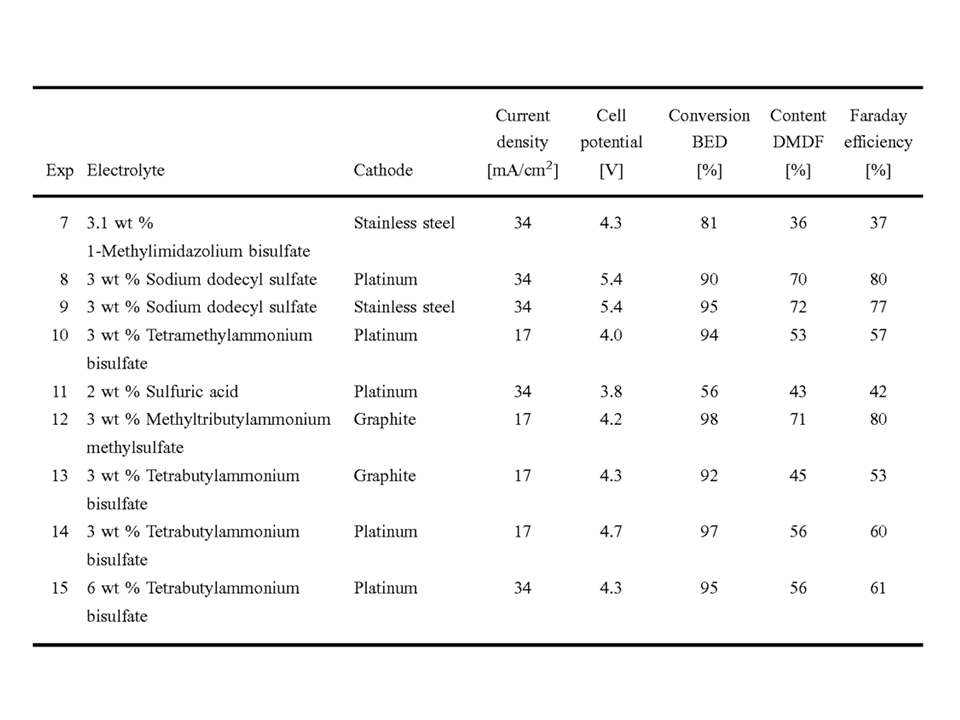
Several quaternary ammonium salts were used as electrolytes and what caught my eye was the fact that the performance of the various quat salts were different, even though other factors were also being changed, so it wouldn’t be fair to try to derive firm conclusions for structure-activity relationships. Structure-activity performance of quats has always been my focus since the late 1970’s.
What I can’t explain (and would like to request your input) is why some of these quats work so well, while others, not so much.
When methyl tributyl ammonium outperforms tetrabutylammonium in classical PTC applications, that usually indicates a “T-reaction.” However, this application is not PTC, it’s an electrochemical oxidation.
Using the same cathode and the same current density, methyltributyl ammonium delivers higher selectivity and higher Faraday efficiency than tetrabutylammonium. While there is a difference in the quat cation, I do not pretend to understand the difference in performance.
Maybe one of our readers has expertise in electrochemistry and can provide some insight about why we see these differences in activity. Is it ion-pairing, local viscosity effects, specific adsorption at the electrode? I have no clue.
I also can’t say that I understand the actual organic reaction, in particular, where does the methoxy group come from in DMDF? Is it possible that under anodic conditions, methanol can form methoxy radicals (·OCH₃) or methoxide ions, which then attack oxidized forms of the diol?
It will be fun to see what our audience comes up with to explain these results.