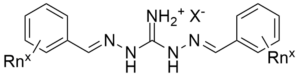
Hexaalkylguanidinium salts are used as thermally stable phase-transfer catalysts. While the guanidinium salts shown in the diagram are not a hexaalkylguanidinium salt, it caught our attention because these compounds have an unusual and surprisingly high affinity for sulfate over chloride.
This is a big deal both in concept and in practice.
Following are two excerpts from the patent Williams; N., Custelcean; R., Seipp; C., Moyer; B., Ellis; R., Abney; C. (UT-Battelle) US Patent 11,001,554, 11-May-2021: “The above described DIG [di-iminoguanidinium] receptor is remarkable in both the unparalleled high selectivity for sulfate over chloride as well as the unique ability to solubilize the superhydrophilic ion pairs in the aliphatic hydrocarbon solvent.” Another statement about the utility of this discovery is “The removal of superhydrophilic anions, such as sulfate and phosphate, from brines, agricultural runoff, and industrial waste continues to be an ongoing challenge.”
The inventors compared these lipophilic di-imino-guanidinium [“DIG”] salts to Aliquat 336 in terms of their abilities to extract sulfate from an aqueous stream. The DIG’s were able to extract 4-5 orders of magnitude more of sulfate into dichloroethane than Aliquat 336 and an additional 1-3 orders of magnitude into hydrocarbons.
Similarly, the lipophilic DIG’s were about 1,000 times more selective toward sulfate than chloride versus Aliquat 336 (dichloroethane as solvent). In hydrocarbons, lipophilic DIG’s were about 1,000-5,000 times more selective for sulfate than chloride. This is very unusual behavior for PTC experts used to opposite behavior for quaternary ammonium cations. Classical phase-transfer catalysts greatly prefer monoanions over dianions and trianions.
It should be noted that the DIG’s for which data were presented apparently have alkyl groups on the aromatic rings that have between 4 and 12 carbon atoms. These extra carbon atoms (Rnx) impart the added lipophilicity to the guanidinium salts. It is not clear what are the exact structures of the DIG’s for which data are shown in the figures.
The patent addresses the thermodynamic factors that are relevant to the classical understanding of why hydration of sulfate versus hydration of chloride makes it so hard to extract sulfate from aqueous solution in the presence of chloride. This is an interesting and surprising patent to those few of us who have been teaching quat-anion affinities for decades.